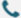As we evaluate experimental design strategies at Synbio Technologies, questions such as qPCR Probe Fluorescence Signal behavior and how can I improve qPCR precision? often guide the way we optimize our workflows. Multiplex qPCR has become valuable for laboratories aiming to extract more information from a single reaction, yet the arrangement of each Fluorophore and Quencher pair directly affects the clarity of the resulting data. When multiple dyes emit in adjacent wavelength ranges, even subtle spectral overlap can influence interpretation. By examining how multiplex configurations alter fluorescence patterns, we gain insights that help us refine probe selection and overall reaction setup.

Understanding How Multiplexing Alters Fluorescence Distribution
Multiplexing relies on different reporter dyes emitting at distinguishable wavelengths so multiple genetic targets can be monitored at once. When we analyze fluorescence curves, we often observe interaction between channels when excitation and emission spectra fall too closely together. This interaction can distort the qPCR Probe Fluorescence Signal, making it appear stronger or weaker than expected. These changes also contribute to inconsistent outcomes that lead researchers to ask how can I improve qPCR precision? across varying templates and concentrations. By focusing on the characteristics of each Fluorophore and Quencher pair—such as their emission shape, environmental sensitivity, and quenching efficiency—we can design reaction sets that minimize cross-channel interference while supporting accurate amplification monitoring.
Practical Considerations for Selecting Dye–Quencher Combinations
In practice, avoiding excessive spectral overlap involves reviewing the full emission range of each dye, especially when four or more targets are evaluated in the same reaction. We routinely assess whether the emission from one dye bleeds into the detection window of another, and how this bleed-through affects the qPCR Probe Fluorescence Signal at different cycle numbers. These evaluations help us explain how can I improve qPCR precision? during multiplex development, as careful probe placement prevents misinterpretation of early amplification signals. The interaction between a Fluorophore and Quencher is also important because quenching efficiency determines baseline noise and affects channel separation. Our internal assessments draw on the same principles described in our resource on factors shaping probe fluorescence, ensuring the guidance we apply aligns with practical laboratory conditions.
Conclusion: Measuring and Managing Spectrum Overlap in Multiplex qPCR
Understanding spectral behavior is essential when multiple fluorescence channels operate simultaneously. By examining how multiplex design affects the qPCR Probe Fluorescence Signal, we gain clearer insight into how can I improve qPCR precision? during assay development and optimization. Monitoring the characteristics of each Fluorophore and Quencher helps us maintain clean signal separation and reduce analytical uncertainty. At Synbio Technologies, we continue to refine these evaluation methods so our workflows support accurate multiplex qPCR experiments from initial planning through final data interpretation.
 DNA Synthesis
DNA Synthesis Vector Selection
Vector Selection Molecular Biology
Molecular Biology Oligo Synthesis
Oligo Synthesis RNA Synthesis
RNA Synthesis Variant Libraries
Variant Libraries Genome KO Library
Genome KO Library Oligo Pools
Oligo Pools Virus Packaging
Virus Packaging Gene Editing
Gene Editing Protein Expression
Protein Expression Antibody Services
Antibody Services Peptide Services
Peptide Services DNA Data Storage
DNA Data Storage Standard Oligo
Standard Oligo Standard Genome KO Libraries
Standard Genome KO Libraries Standard Genome Editing Plasmid
Standard Genome Editing Plasmid ProXpress
ProXpress Protein Products
Protein Products