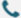As Synbio Technologies, we understand that accuracy in gene sequences is essential for research, development, and production across modern applications. As a Synthetic Biology Company, we work with teams who expect reliable design methods and transparent quality control processes. When researchers prepare a synthetic biology product or investigate a new synthetic biology therapeutic, they rely on a foundation of correct sequences, validated materials, and consistent workflows. For this reason, we continue refining internal systems to maintain dependable results as projects grow in complexity.
Building Accurate Sequences Through Rigorous Design
Ensuring accuracy begins with well-structured digital design. As a Synthetic Biology Company, we use established analytical checks to confirm reading frames, codon usage patterns, and structural feasibility before the gene reaches the build stage. These steps reduce downstream revision cycles and help teams preparing a synthetic biology product plan experiments with fewer unexpected obstacles. When projects involve early research for a synthetic biology therapeutic, our approach supports a smooth transition from conceptual design to physical sequence construction. To make this process accessible, we offer tools that guide users through DNA planning, allowing them to verify design assumptions while maintaining full control of their scientific direction. Our internal experience and manufacturing background ensure that design choices remain aligned with stable production practices.
Implementing Quality Control Across Construction and Validation
Sequence accuracy depends not only on design but also on careful assembly and verification. As we prepare each construct, we apply controlled workflows that include stepwise monitoring and verification checkpoints. Because we operate as a Synthetic Biology Company, we commit to using validated processes that minimize variation and maintain dependable outcomes. These systems support researchers designing a synthetic biology product for industrial testing or exploring components relevant to synthetic biology therapeutic development. Quality control includes sequence confirmation, impurity analysis, and documentation that clarifies how each construct was prepared. These methods allow users to integrate our materials confidently into larger experimental pipelines. Through these measures, we aim to help scientific teams reduce rework and preserve momentum across fast-moving projects.
Supporting Reliable Application Through Transparent Delivery
After validation, accurate documentation and delivery practices ensure users can apply the sequences effectively. Many teams working on a synthetic biology product require consistent batches, predictable formatting, and clear record-keeping. Others exploring a synthetic biology therapeutic need sequence traceability across multiple iterations. As a Synthetic Biology Company, we support these expectations through structured logistics and communication. Our experience in the design-build-test-learn framework allows us to provide materials that suit research timelines and industrial workflows. This approach is strengthened by our background in DNA synthesis and related service development, which we describe openly on our company information platform, ensuring users understand how our systems operate and how materials are produced.
Conclusion: Creating Confidence in Every Synthetic Gene
Reliable performance in synthetic gene projects depends on intentional design, structured validation, and transparent delivery. As Synbio Technologies, we work to provide clear processes that help users maintain accuracy across projects of different scales. Whether preparing a synthetic biology product or exploring early components for a synthetic biology therapeutic, we apply consistent practices that reflect our role as a Synthetic Biology Company committed to dependable sequence quality.
 DNA Synthesis
DNA Synthesis Vector Selection
Vector Selection Molecular Biology
Molecular Biology Oligo Synthesis
Oligo Synthesis RNA Synthesis
RNA Synthesis Variant Libraries
Variant Libraries Genome KO Library
Genome KO Library Oligo Pools
Oligo Pools Virus Packaging
Virus Packaging Gene Editing
Gene Editing Protein Expression
Protein Expression Antibody Services
Antibody Services Peptide Services
Peptide Services DNA Data Storage
DNA Data Storage Standard Oligo
Standard Oligo Standard Genome KO Libraries
Standard Genome KO Libraries Standard Genome Editing Plasmid
Standard Genome Editing Plasmid ProXpress
ProXpress Protein Products
Protein Products