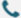Naturally occurring heavy-chain antibodies in alpaca blood lack light chains and consist of a single variable domain (VHH) and two constant domains (CH2 and CH3)[1]. Unlike engineered single-chain variable fragments (scFvs), these antibodies do not readily self-aggregate. When the VHH domain is cloned and expressed on its own, it remains structurally stable and retains antigen-binding activity comparable to the full-length heavy-chain antibody.
(a) The structure diagram of the conventional antibody
(b) Schematic diagram of nano-structure
This VHH, the smallest functional antigen-binding unit—about 15 kDa and one-tenth the size of a conventional antibody,is known as a Nanobody (Nb). With its simple architecture, high stability, and strong binding affinity, the Nanobody represents a major innovation in antibody technology.
Nanobodies can be efficiently produced in engineered bacteria rather than hybridoma cells, making them easy to express and genetically modify. Their stability and excellent tissue penetration have made them highly attractive for applications in therapeutics, diagnostics, and molecular detection.
What is the advantage ofnanobodies ?
01 High Affinity and Miniaturization
Despite their significantly smaller size compared to conventional antibodies, Nanobodies exhibit uncompromised antigen-binding activity. This is primarily attributed to their unique structure: their CDR3 loops are often longer, effectively compensating for the loss of the binding interface caused by the absence of light chains[2]. Furthermore, Nanobodies lack superfluous structural elements, retaining only the essential antigen-binding site, which is encased within conserved framework regions to form distinctive protruding or pocket-like cavities. This architecture enables Nanobodies to precisely recognize and bind to hidden or recessed epitopes that are often inaccessible to conventional antibodies, thereby achieving "small size yet high activity."
02 Superior Stability
The presence of additional intra-domain disulfide bonds confers exceptional conformational stability to Nanobodies, making them highly resistant to inactivation under varying environmental conditions. Consequently, they demonstrate remarkable tolerance to high temperatures, extreme pH levels, and organic solvents. Studies have shown that some Nanobodies can remain stable for several months, or even up to a year, at 37°C[3]. This characteristic enables room temperature transportation and storage, overcoming the cold chain requirements essential for conventional antibodies, and also underscores their excellent capacity for reversible renaturation.
03 Excellent Solubility
Throughout their evolution, key hydrophobic amino acids in the FR2 region of Nanobodies have been replaced by hydrophilic residues. This structural refinement, combined with the coverage of some hydrophobic patches by their extended CDR3 loops, significantly enhances the molecule's hydrophilicity and effectively prevents aggregation and dimerization[4]. As a result, Nanobodies typically exhibit high solubility and can be efficiently produced as soluble proteins in prokaryotic expression systems.
04 Powerful Tissue Penetrability
Owing to their minimal molecular dimensions, Nanobodies can rapidly traverse tissue barriers and even enter cells to efficiently engage intracellular targets. This superior penetration capability is a clear advantage over the substantially larger conventional antibodies, laying a solid foundation for their applications in targeted therapies and intracellular imaging.
A Next-Generation Tool for Therapy and Diagnosis
Nanobodies (VHH), leveraging their small size, high stability, and ease of engineering, are reshaping diagnostic and therapeutic paradigms in oncology, neurology, and infectious diseases[5].
1. Cancer Diagnosis and Therapy
Tumor Imaging: Nanobodies can be radiolabeled as molecular probes for high-contrast PET/SPECT imaging within hours post-injection, enabling earlier tumor detection, improved staging accuracy, and real-time monitoring.
Targeted Drug Delivery: Acting as precise “targeting modules,” Nanobodies can be conjugated to drugs, toxins, or radionuclides to enhance delivery into solid tumors, improve therapeutic outcomes, and reduce systemic toxicity.
Immune Checkpoint Modulation: Their highly engineerable structure supports the development of novel immunomodulatory agents that act on pathways such as PD-1/PD-L1.
2. Cell Therapy: Advancing Precision Medicine
CAR-T/CAR-NK Engineering: Incorporating Nanobodies as the antigen-binding domains in CAR constructs can enhance CAR stability and expression efficiency. Their extended CDR3 loops allow recognition of cryptic epitopes, broadening the repertoire of targetable antigens.
Bi-/Multi-specific Antibodies: Nanobodies can function as modular components for building multi-specific molecules that engage both tumor cells and immune cells, thereby amplifying anti-tumor activity.
3. Neuroscience: Breaching the Blood-Brain Barrier
With their small size and high biochemical stability, Nanobodies have the potential to penetrate the blood–brain barrier, offering new opportunities for diagnosing and treating central nervous system disorders including Alzheimer’s disease, Parkinson’s disease, and brain tumors.
4. Infectious Disease Prevention and Control
Antiviral Applications: Nanobodies can bind conserved viral epitopes, delivering potent neutralization while lowering the risk of escape mutations. They show strong potential against emerging pathogens such as SARS-CoV-2[6], influenza, and Ebola, with advantages including rapid development timelines, superior stability, and the possibility of inhalable formulations for respiratory delivery.
Antibacterial Applications: Nanobodies can be engineered for rapid diagnosis of bacterial infections or used as targeted enhancers to boost the efficacy and specificity of antimicrobial agents.
Synbio Technologies’ Nanobody Services
Leveraging the broad application potential of nanobodies, Synbio Technologies offers mature and highly efficient end-to-end nanobody customization services. Our comprehensive workflow includes antigen preparation, alpaca immunization, antibody library construction, nanobody screening, as well as expression and purification. We provide integrated solutions to support clients in nanobody development and production.
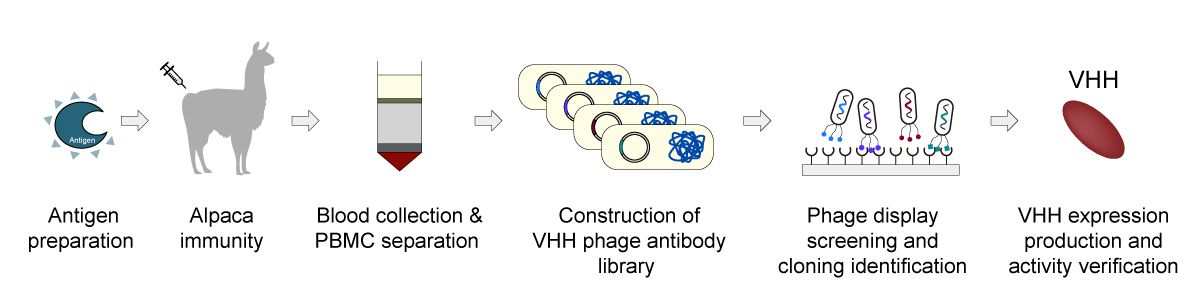
Core Advantages
-
Source Control: We operate our own alpaca ranch, ensuring fully traceable, high-quality, and naturally non-immunized starting materials.
-
Proven Expertise: Each step from immunization to library construction and screening is supported by strict QC and extensive project experience.
-
End-to-End Delivery: Provide your target or antigen, and we manage the entire workflow from immunization to sequencing verification and protein delivery, significantly reducing your R&D time.
-
Customized Services: We tailor development strategies to your application needs, delivering Nanobody molecules optimized for each use case.
Nanobodies represent not merely an incremental improvement in antibody format, but a key to unlocking new possibilities in precision medicine, advanced diagnostics, and fundamental research. With our professional platform and efficient services, Synbio Technologies is your reliable partner in exploring the vast potential of the Nanobody world.
References
[1]Wang J, Tong T, Wu Q. Nanobodies in animal infectious disease control: diagnosis and therapy. Front Cell Infect Microbiol. 2025 Jul 25;15:1640352.
[2]Cai H, Yao H, Li T, et al. An improved fluorescent tag and its nanobodies for membrane protein expression, stability assay, and purification. Commun Biol, 2020, 3(1): 753
[3]Akazawa-Ogawa Y, Takashima M, Lee Y H, et al. Heat-induced irreversible denaturation of the camelid single domain VHH antibody is governed by chemical modifications. J Biol Chem, 2014, 289(22): 15666-15679
[4]Melarkode Vattekatte A, Shinada N K, Narwani T J, et al. Discrete analysis of camelid variable domains: sequences, structures, and in-silico structure prediction. Peer J, 2020, 8: e8408
[5]Muyldermans S. A guide to: generation and design of nanobodies. FEBS J. 2021 Apr;288(7):2084-2102.
[6]Liu, Q., Lu, Y., Cai, C. et al. A broad neutralizing nanobody against SARS-CoV-2 engineered from an approved drug. Cell Death Dis 15, 458 (2024).
 DNA Synthesis
DNA Synthesis Vector Selection
Vector Selection Molecular Biology
Molecular Biology Oligo Synthesis
Oligo Synthesis RNA Synthesis
RNA Synthesis Variant Libraries
Variant Libraries Genome KO Library
Genome KO Library Oligo Pools
Oligo Pools Virus Packaging
Virus Packaging Gene Editing
Gene Editing Protein Expression
Protein Expression Antibody Services
Antibody Services Peptide Services
Peptide Services DNA Data Storage
DNA Data Storage Standard Oligo
Standard Oligo Standard Genome KO Libraries
Standard Genome KO Libraries Standard Genome Editing Plasmid
Standard Genome Editing Plasmid ProXpress
ProXpress Protein Products
Protein Products