Monoclonal antibody sequencing includes hybridoma cell line sequencing and clonal B cell sequencing. Each of these kinds of cells can produce a single antibody against a unique antigen after screening. Synbio Technologies uses both Sanger sequencing and NGS sequencing technologies to provide both accurate and efficient hybridoma/clonal B cell sequencing services across a variety of species. We provide a fast and easy way to sequence your hybridoma antibodies, which will help you identify the best therapeutic leads for future drug development.
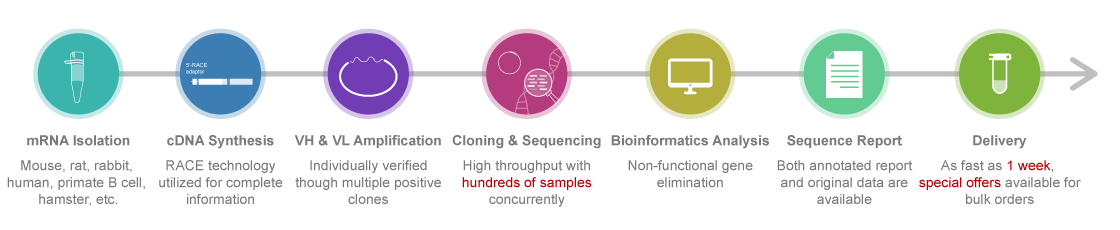
Standard Hybridoma Sequencing Process
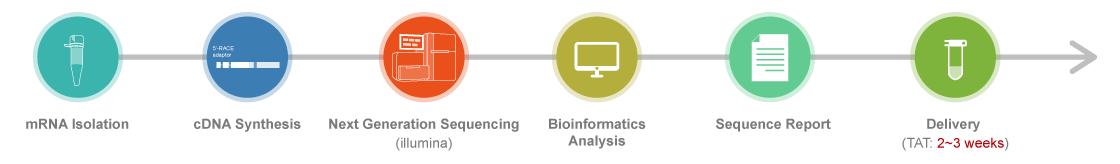
NGS Sequencing Process
The Synbio Tech Difference
- Comprehensive: Sequencing of heavy and light variable regions or full-length of antibody sequences.
- Wide applicability: Relying on a professional immunogenetics database and our rich experience in antibody sequence analysis and design, antibodies from any species (mouse, rat, human, rabbit, hamster, camel, sheep, etc.) and various antibody subtypes (IgG1, IgG2a, IgG2b, IgG3, IgG4, IgM, IgE, IgA etc.) can be designed and sequenced.
- Reliability: At least 4 positive clones are selected for Sanger sequencing and sequence analysis is performed to exclude any invalid sequences.
- High-throughput + great savings: Whether one or many samples, we can do it all. We provide volume discounts; meaning, the more samples you provide, the more money you will save!
- Fast turnaround time: We provide express sequencing services, as fast as one week.
- One-stop service: We provide a one-stop solution from hybridoma sequencing to recombinant antibody production. For the non-human antibodies, our antibody humanization service can be considered.
Cell Sample Preparation and shippping
- For hybridoma cell lines or clonal B cells: customers will need to submit hybridoma cell samples (>1×106 cells) and indicate the species and subtype of the monoclonal antibody.
| Sample | Shipping |
|---|---|
| Cells in RNAlater or DNA/RNA Shield (Recommended) |
Ice pack (Domestic)
Dry ice (International) |
| Cell pellets | Dry ice |
| Cells in cryopreservation media | Dry ice |
| Good quality total RNA | Dry ice |
Service Specifications
| Method | Price | TAT | Deliverables |
|---|---|---|---|
| Sanger sequencing | Starting from $800 | As fast as 5 business days |
|
| NGS | Starting from $2,000 | 2 – 3 weeks |
|
Which method is right for your research?
In addition to carefully weighing cost and turnaround time considerations, selecting the optimal sequencing method for your project also requires a thoughtful consideration of your overall research goals. If your objective is to identify the complete length of the antibody, the sanger sequencing approach may prove to be the most fruitful strategy. However, if you are looking to detect low abundance variants, NGS technology is almost certainly the way to go.
By sequencing the complete length of the antibody, sanger sequencing delivers highly precise results, allowing for the identification of even small variances that might otherwise be missed. Meanwhile, NGS enables the detection of rare variants that may be present at very low levels, which can be important for applications where the identification of diverse samples is critical.
Contact our team to share about your project, and our experts provide insight and support throughout the sequencing process.
Applications
Monoclonal antibody sequencing is an essential tool in the biotechnology industry that provides multiple benefits and applications for research and development. Here are some of the ways in which accurate antibody sequencing can help researchers in their endeavors:
- Helps identify and define the attributes of antibodies: Monoclonal antibody sequencing can provide valuable insights into the characteristics of the antibody molecule, such as its specificity, binding affinity, and structural elements. These insights can help researchers optimize their monoclonal antibody development and manufacturing processes.
- Supports patent applications: Accurate sequencing of monoclonal antibodies can aid researchers in protecting their intellectual property and securing patent protection. By providing detailed information on the composition and structure of the antibody, patent applications can be strengthened, and potential challenges can be avoided.
- Prevents cell line loss: Monoclonal antibody sequencing can also facilitate the permanent storage of hybridoma cell lines and prevent the loss of valuable monoclonal antibody-producing cells. This is especially crucial for long-term research projects that require consistent access to the same cell lines.
- Aids in the selection of appropriate cell lines: Sequencing monoclonal antibodies can help researchers identify the ideal cell lines best suited for the development of recombinant monoclonal antibodies. With this knowledge, researchers can select the best cell line expression systems and generate monoclonal antibodies with higher purity.
- Accelerates the generation of monoclonal antibodies: Monoclonal antibody sequencing can shorten the period of hybridoma cloning and accelerate the generation of monoclonal antibodies. This can help save time, resources, and money, making the antibody development process more efficient and streamlined.
- Affinity maturation: Sequencing can also aid in affinity maturation to further enhance the affinity of monoclonal antibodies. This process involves iterative rounds of mutation and selection to optimize the antibody’s binding affinity to the target, making it more effective in treating the intended disease.
- Build chimeric and humanized antibodies: Monoclonal antibody sequencing can serve as a foundation for developing chimeric and humanized antibodies. By determining the structure and composition of the monoclonal antibody, researchers can modify it, optimizing its therapeutic properties and reducing potential side effects. These modifications can allow for the development of more effective and safer treatment options for patients.
Standard Antibody Variant Region PCR Amplification Primers Design
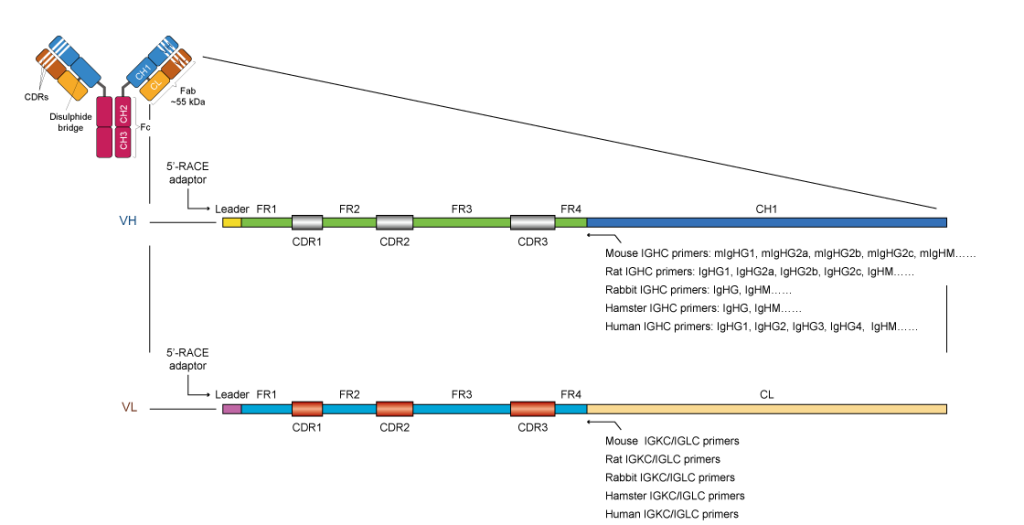
mRNA isolated from hybridoma samples is subjected to RT-PCR with unique species-specific primer sets to amplify the target regions. Synbio Technologies’ antibody variable region sequencing service has identified more than 10,000 hybridoma cell lines. More than 95% of the samples have been successfully sequenced with both the heavy chain and light chain identified. Even with technically difficult samples, Synbio Technologies’ approach has demonstrated a high success rate in numerous cases. Synbio Technologies’ service, coupled with online data tracking and management system, provides customers with absolute confidence at every step.
Following the successful cloning of variable regions, the sequences can be fused to a variety of IgG or IgM backbones. Alternatively, variable region sequences can be reformatted as scFv or bispecific antibodies in various configurations. Transiently produced antibodies can then be validated and characterized by downstream analytical services.

