Imagine a world where your medication is tailored just for you. This isn’t science fiction – it’s the promise of mRNA technology. By turning cells into miniature drug factories, mRNA is changing the game in treating a variety of diseases. With breakthroughs in understanding its structure and function, we’re on the brink of a healthcare revolution. This is where the incredible journey of mRNA technology, leading the charge in cell and gene therapy, begins.
The Key Domains of mRNA and Function
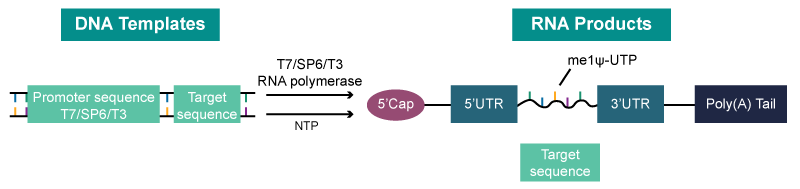
mRNA, or messenger RNA, is like the postman of the cellular world, delivering vital genetic instructions. It’s a single-stranded RNA molecule that plays a crucial role in protein synthesis. Imagine mRNA as a string of important text messages, each part playing a unique role:
- 5’ Cap: Think of this as the ‘security badge’ for mRNA. It helps initiate the process and protects the mRNA from being degraded.
- UTR: These are like the regulatory notes at the beginning and end of our messages, controlling how the main content is handled.
- ORF: This is the heart of the message, the essential part that dictates the protein’s creation. Optimizing this segment is like tweaking a message to avoid misunderstandings.
- Poly(A) Tail: Similar to a postscript in a letter, this part helps stabilize the mRNA and plays a role in how the message is read.

The Mechanism of In Vitro Transcription
In our quest to conquer disease, we often turn to nature for answers. In this case, it’s RNA – a key player in gene regulation. Here’s a simplified breakdown of how we harness RNA in the lab:
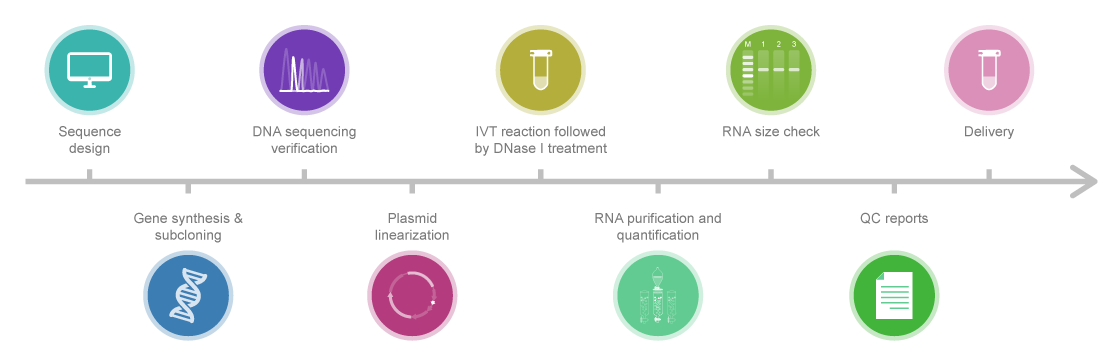
- Preparing High-Quality Transcription Templates: It’s all about getting the blueprints right.
- IVT mRNA Capping Methods: Think of this as putting the right hat on our mRNA – a crucial step for it to be recognized and functional.
- Nucleotide Modification of IVT mRNA: This is about making our synthetic mRNA look like the body’s own to avoid immune responses.
- IVT mRNA Tail Addition Method: Adding the ‘tail’ to our mRNA – a bit like determining the length of a message.
- IVT mRNA Purification: This step is like filtering out typos and errors from our message.
- IVT mRNA Quality Control: Ensuring our message is clear, concise, and correctly formatted.
The potential of IVT mRNA is enormous – it’s like giving cells a blueprint to produce almost any protein. Unlike DNA drugs, mRNA drugs pose no risk of integrating into our DNA and can reach places traditional drugs can’t. As interest in RNA research grows, so does the demand for top-notch IVT mRNA synthesis. At Synbio Technologies, we’re not just participating in this revolution – we’re driving it. Our one-stop IVT mRNA solution, from template synthesis to extensive quality control, is setting new standards in the industry.
Eager to explore the potential of mRNA technology for your research or product development? Contact Synbio Technologies today and discover how our expertise can accelerate your R&D journey.
References
[1] Sun H, Zhang Y, Wang G, Yang W, Xu Y. mRNA-Based Therapeutics in Cancer Treatment. Pharmaceutics. 2023 Feb 13;15(2):622. doi: 10.3390/pharmaceutics15020622. PMID: 36839944; PMCID: PMC9964383.
[2] Zhang J, Liu Y, Li C, Xiao Q, Zhang D, Chen Y, Rosenecker J, Ding X, Guan S. Recent Advances and Innovations in the Preparation and Purification of In Vitro-Transcribed-mRNA-Based Molecules. Pharmaceutics. 2023 Aug 23;15(9):2182. doi: 10.3390/pharmaceutics15092182. PMID: 37765153; PMCID: PMC10536309.
[3] Kang DD, Li H, Dong Y. Advancements of in vitro transcribed mRNA (IVT mRNA) to enable translation into the clinics. Adv Drug Deliv Rev. 2023 Aug;199:114961. doi: 10.1016/j.addr.2023.114961. Epub 2023 Jun 14. PMID: 37321375; PMCID: PMC10264168.