The Pivotal Role of Collagen
Discover the secret to age-defying with collagen, the ultimate core element of anti-aging that minimizes the effects of aging in the body. Collagen proteins, with 28 unique members, are crucial in providing strength, elasticity, and structural integrity to skin, bones, and tendons. Not only does collagen boast an impressive lifespan and mechanical strength, but it also forms fibrils and networks, making it a sought-after material in biomedicine, drug and food industries, and cosmetics.
Methods to Produce Collagen
Collagen, a complex protein, can be produced through two primary methods:
- Extraction from Animal Sources: Collagen is extracted from animal tissues (e.g., pig, donkey, cow, fish) using acid-base and enzymatic treatments.
- Recombinant Collagen Technology: This technology involves engineering human collagen genes with specific sequences and transferring them into host cells, which can include microorganisms, animals, and plants. The collagen genes are then fermented to induce the expression of collagen or its analog.
Comparison of Natural Collagen and Recombinant Human-like Collagen
| Types | Natural Collagen | Recombinant Human-like Collagen |
| Production Technology | Chemical Extraction | Biofabrication |
| Affinity | Weak | High |
| Allergenicity | High Allergy Risk | Low Allergy Risk |
| Security | Vulnerable to Animal Viruses | Virus-free |
| Biological Activity | Low | High |
| Purity | Mixed Collagen, Complex Composition |
Single collagen, Fixed Composition, ≤ 95% purity |
The Superiority of Recombinant Collagen
While animal-derived collagens can be purified in large amounts at a relatively low cost, the method may carry viruses, posing a safety hazard. Recombinant collagen, on the other hand, offers strong water solubility, processability, and stable quality. It addresses the safety issues of animal-derived collagens and enhances performance by reducing immune rejection. Moreover, it significantly diminishes the need for animal farming and its associated environmental impacts, such as deforestation and greenhouse gas emissions.
Market Trends and Projections
Pioneering experiments from three decades ago ignited the exploration of recombinant human collagens, potentially replacing animal-derived collagens. According to Grand View Research, the global collagen market is anticipated to reach $17.258 billion in 2022 and is projected to climb to $22.622 billion by 2027, growing at a CAGR of 5.42%.
Challenges and Solutions in Recombinant Collagen Production
The production process of recombinant collagen encompasses expression system construction, fermentation, and purification. However, due to the challenges and high costs associated with cultivating animal and plant cells, utilizing microorganisms like E. coli and yeast as hosts is becoming an industrial trend. Technical difficulties in collagen extraction via genetic engineering include synthesizing the target gene, constructing the triple helix structure, cell transfection, and protein purification.
The Significance of Triple-Helical Collagen Molecules
Triple-helical collagen molecules’ ability to assemble into supramolecular structures underpins their commercial use in the food industry and medical applications, such as cosmetic surgery and tissue repair. When preparing a triple-helical peptide with a specific collagen sequence, it’s vital to consider the peptide’s triple-helix stability. Altering collagen sequences may cause the collagen to lose its essential functions, indicating that the correct amino acid sequence is crucial for proper collagen functionality.
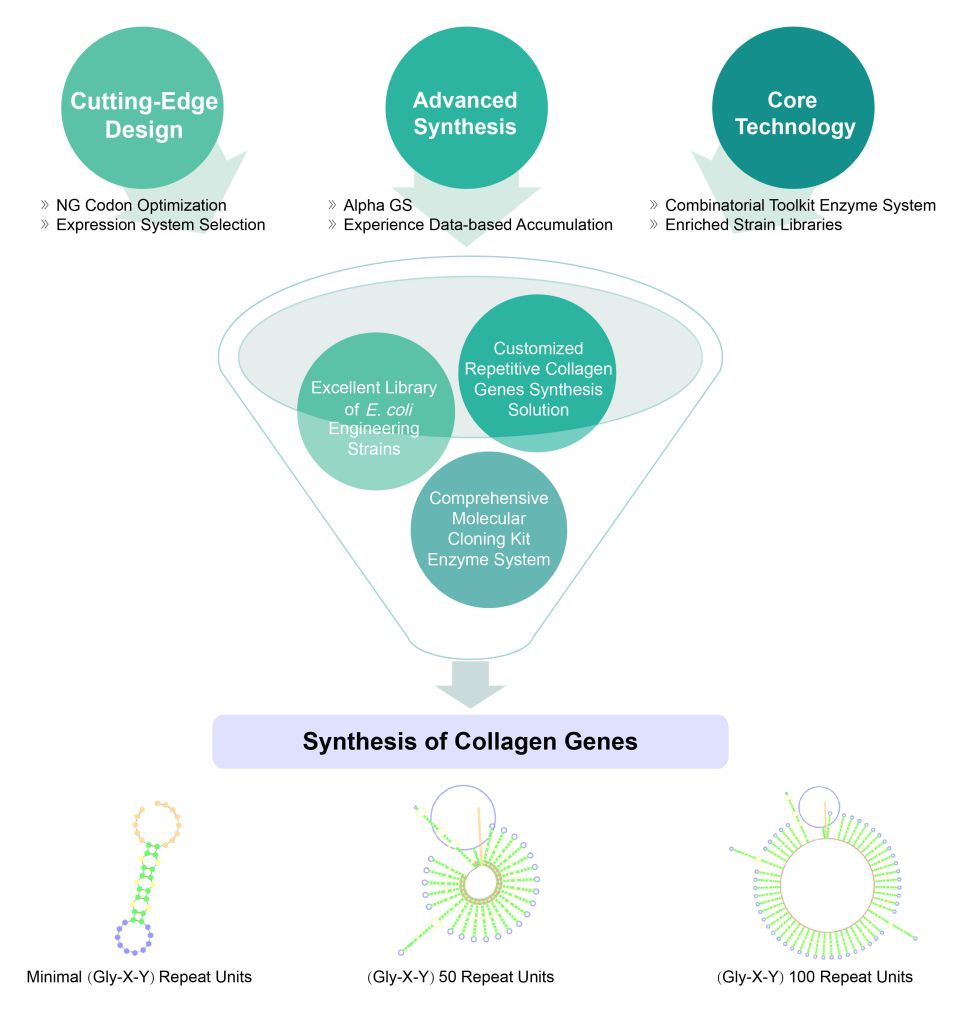
Synbio Technologies: Your Partner in Recombinant Collagen Synthesis
Synbio Technologies has extensive experience in the gene synthesis of recombinant collagen. Our combination of cutting-edge design, advanced manufacturing, and core intelligence tools ensures accurate synthesis of highly repetitive amino acid sequences for recombinant collagen. Our customized synthetic design solution addresses source gene difficulties, opening up possibilities for your collagen research.
Accelerating Production Cycles with Advanced Technology
At Synbio Technologies, we facilitate faster, more efficient production cycles through our advanced synthetic bio-enabling technology platform. Experience intelligent, accurate, and rapid DNA synthesis with our innovative tools: Complex Index (CI), NG Codon, AI-TAT, and DNA Studio bio-intelligent analysis. Engage in extensive DNA design in the lab to optimize protein expression and unlock collagen’s potential in medical, food, and biomedical applications.
Looking Ahead: The Future of Biological Research
According to a report from the Wall Street Journal on September 19, 2023, “Artificial intelligence is pushing the frontiers of biological research, helping scientists program organisms like software engineers write code.” This acceleration in synthetic biology, driven by AI, is swiftly moving from the lab to the marketplace. In the future, with “gene regulation” and “engineering design” at the core, we will initiate from the directional design of collagen molecules and the construction of cell factories. Through design, construction, and test optimization, we aim to break through natural evolution limitations and anticipate the optimization of high-yield expression and large-scale production of collagen under AI design guidance.
The advancements in recombinant collagen gene synthesis signify a pivotal shift towards sustainable biomaterial production. With the integration of AI and synthetic biology, the future holds promising possibilities for optimizing collagen production, enhancing its functionalities, and expanding its applications across various industries.
References:
- Sorushanova A, Delgado LM, Wu Z, Shologu N, Kshirsagar A, Raghunath R, Mullen AM, Bayon Y, Pandit A, Raghunath M, Zeugolis DI. The Collagen Suprafamily: From Biosynthesis to Advanced Biomaterial Development. Adv Mater. 2019 Jan;31(1):e1801651. doi: 10.1002/adma.201801651.
- Ricard-Blum S. The collagen family. Cold Spring Harb Perspect Biol. 2011 Jan 1;3(1):a004978. doi: 10.1101/cshperspect.a004978.
- Fertala A. Three Decades of Research on Recombinant Collagens: Reinventing the Wheel or Developing New Biomedical Products? Bioengineering (Basel). 2020 Dec 2;7(4):155. doi: 10.3390/bioengineering7040155.
- Koide T. Designed triple-helical peptides as tools for collagen biochemistry and matrix engineering. Philos Trans R Soc Lond B Biol Sci. 2007 Aug 29;362(1484):1281-91. doi: 10.1098/rstb.2007.2115.